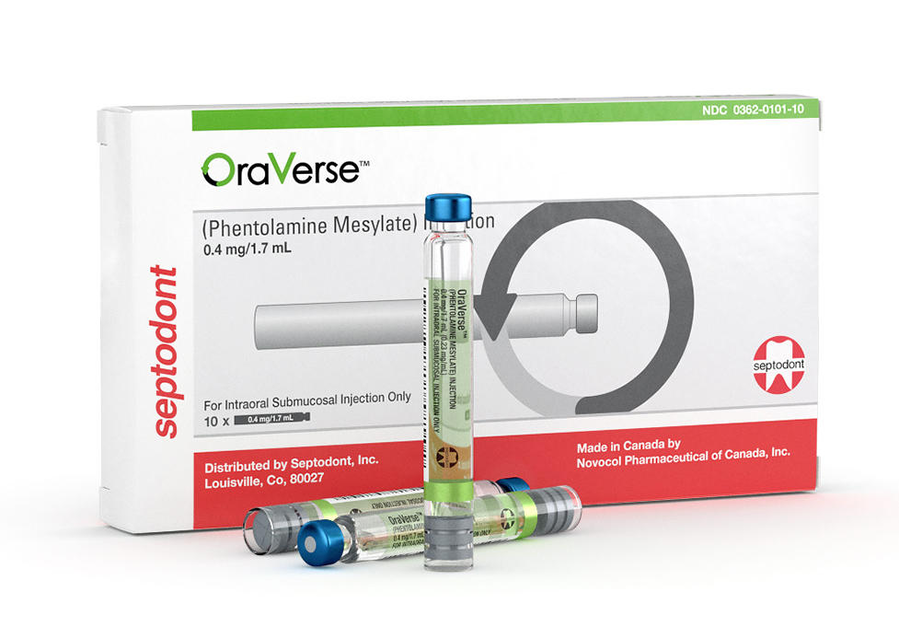
OraVerse update: 10 years and going strong!
October 30 2019
There’s still only one reversing agent in dental anesthetics, and it’s OraVerse, from Septodont/Novocol. We stock it, of course. Our customers love it. It’s hard to find a dental professional who won’t use it, and it’s hard to find a patient who doesn’t want it.
We note that there’s been a fair bit of new trial evidence about the drug over the last few years, and it’s worth a quick mention, just to keep our readership up-to-date about what’s known about who can use OraVerse, whether there are adverse events yet, and so on. The truism is that you don’t really know the value of a drug until it’s been out for a decade, being used in the real world. In the case of OraVerse, all the update news actually seems very good.
OraVerse is phentolamine mesylate, or phenol, 3-[[(4,5-dihydro-1H-imidazol-2-yl)methyl](4-methyl-phenyl)amino]-,methanesulfonate (salt ), in case you’re interested. That’s C17H19N30•CH403S. It’s a non-specific alpha adrenergic blocker. The manufacturers buffer it with acetic acid or sodium hydroxide.
Phentolamine vasodilates in smooth muscle, which is why it’s for use in reversing vasoconstrictive agents. (That’s all it does – it’s not an antidote for anesthetic overdose.) It’s been a component in other medications since the 1950’s, which is reassuring. It’s 100% bioavailable in submucosal injection, and it works pretty fast for your usual dental patient, getting to Cmax in 10 or 20 minutes in adults, and faster than that in kids. Its half-life is 2-3 hours.
In dental trials, OraVerse has been tested with lidocaine 2% with epinephrine 1:100,000, articaine 4% with epinephrine 1:100,000, prilocaine 4% with epinephrine 1:200,000, and mepivacaine 2% with levonordefrin 1:20,000.
You use OraVerse at the end of whatever procedure you’re doing, when gingival or pulpal anesthetic is no longer necessary. It goes in at very same place, and in the very same way (infiltration or block injection) as the anesthetic did.
When you disinfect the carpule cap, try to use U.S.P. grade isopropyl or ethyl alcohol. Some of the cheaper brands denature rubber. (Not everyone knows that.)
You don’t need to worry about any drug interactions. There still aren’t any known. OraVerse is really only contraindicated in patients who might be sensitive to something in the preparation. We don’t know whether it’s okay for really tiny children, or for pregnant mothers, so all advice is to refrain from using OraVerse for these patients for now.
Are there side effects? Barely. The most common complaint in clinical trials is injection site pain, but the percentages of people who complain are in the single digits, and are nearly identical with controls.
This goes as well for tachycardia, bradycardia, post-operative pain, and headache, where the numbers really are very small. There’s been no record yet of any serious adverse event, even after more than 10 years of clinical use, and there’s been no trial discontinuation due to an adverse event.
Adverse events in trials reported by less than 3% of patients that occurred at a greater incidence than with controls have included diarrhea, facial swelling, hypertension, injection site reactions, mouth or jaw pain, paresthesia, pruritus, upper abdominal pain, and vomiting. These were not only rare, but mostly mild, and resolved within 48 hours.
You may see allusions in literature to infarction, cerebrovascular spasm or occlusion, acute and prolonged hypotensive episodes and cardiac arrhythmias, or dizziness, weakness, or orthostatic hypotension. These have been identified only in parenteral phentolamine. Even here, it was rare. You’d check anyway, but we do see recommendations in literature to be sure about patients’ cardiac history before you give them OraVerse. We note that no deaths have ever been reported, even with parenteral administration.
What to do about adverse events, if they happen? Guidance overall is just to respond in customary fashion to whatever it is. There is no antidote to OraVerse.
OraVerse at recommended doses does not affect fertility in animal models. Data about drug-associated risk for major birth defects or miscarriage in humans do not exist. Pregnant women have been excluded from phentolamine trials thus far. In animal studies, there were skeletal immaturities and decreased growth in offspring at doses at least 24-times the recommended dose, and lower implantation rate with phentolamine at 60-times the recommended dose. No malformations or embryofetal deaths have been observed in the offspring of pregnant mice, rats, or rabbits, even at respective organogenesis doses of 24, 60, and 20 times the recommended doses.
OraVerse has not to our knowledge been studied for carcinogenesis.
Safety and efficacy have still not been established in patients younger than 3 years or weighing less than 15 kg (33 lbs), and use of the drug is therefore not recommended for this group. For patients 3 years and above, the recommendations are to dose according to body weight.
Trials have not so far identified safety or response differences in elderly patients. Advice is just to keep older people’s frailties in mind (as you would anyway).
OraVerse remains a popular product, and it deserves to be. You’ll know from your own experience that patients won’t start aching when you reverse their anesthetic out. This is borne out in trials, though this was for routine restorative and periodontal maintenance procedures, not surgery, or anything, where you’d want more analgesia, not less. OraVerse isn’t for those situations.
But for routine use it works very well. It’s rugged and reliable, and causes no problems to speak of. Here’s a test case, about just how rugged and reliable that is. We note a paper reporting on portable dental care in a rural school system in the United States – which must be the wildest place on earth to be a dentist. In 923 dental visits in this study, where local anesthetic was administered, there was a complication rate, of 5.3%. These were moderate complications only. They were chiefly – remember, these were schoolkids – ‘self-inflicted soft tissue injury’. Phentolamine mesylate, the study found, was determined to improve safety outcomes in these kids. Teachers noticed that the treated children were better able to reorient back to classwork, and were not disruptive to their classmates. OraVerse made life better even in portable school dentistry.
OraVerse is here to stay. It’s effective and safe, and all new new data say so. We’ve got it in stock for you, of course. OraVerse comes in boxes of 10 and 50 cartridges. The 50-cartridge box contains 5 trays with 10 cartridges each. Each cartridge contains 0.4 mg of phentolamine mesylate in 1.7 mL of solution.
Recent posts
07-18-2025
Textured vs. Smooth Nitrile Gloves

02-12-2025
Ultrasonic scalers

10-17-2024
High Definition Black Line Mirrors

09-27-2020